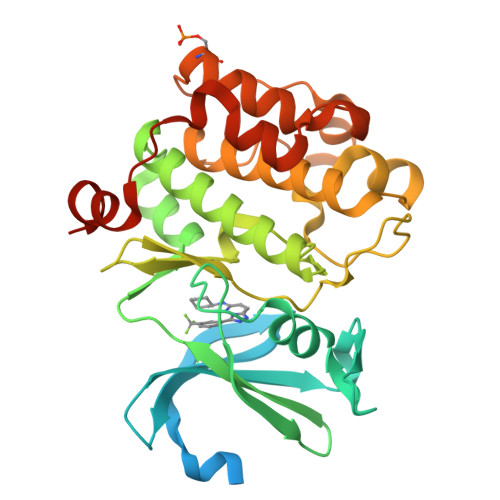
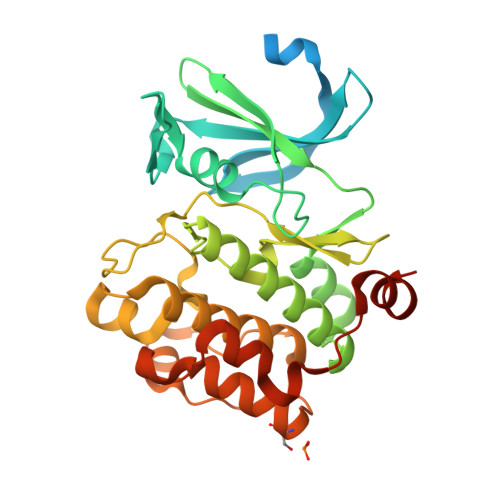
Docking study yields four novel inhibitors of the protooncogene pim-1 kinase.
Pierce, A.C., Jacobs, M., Stuver-Moody, C.(2008) J Med Chem 51: 1972-1975
- PubMed: 18290603
- DOI: https://doi.org/10.1021/jm701248t
- Primary Citation of Related Structures:
3BGP, 3BGQ, 3BGZ - PubMed Abstract:
To supplement the hits from a high throughput screen, docking was performed against Pim-1 kinase. Glide docking was augmented with a filter to require traditional or aromatic CH..O hydrogen bonds to the kinase hinge. Four diverse actives, of 96 molecules assayed, had K(i) values between 0.091 and 4.5 microM. This gives a 14-fold enrichment over the earlier HTS run, and the two crystal structures solved confirmed the binding modes predicted by docking.
Organizational Affiliation:
Vertex Pharmaceuticals Incorporated, 130 Waverly Street, Cambridge, MA 02139, USA. al_pierce@vrtx.com